Explore precision PRP with photobiomodulation and its innovative approach to enhance healing and recovery in medical treatments.
Table of Contents
Introduction
As a clinician deeply invested in orthobiologics, my primary goal has always been to optimize patient outcomes by leveraging the most advanced, evidence-based treatments available. I’m Dr. Alexander Jimenez, and in my practice, I combine chiropractic care with my role as a Family Nurse Practitioner (FNP-APRN) to offer a comprehensive, integrative approach to musculoskeletal health. This educational post distills my clinical experience, insights from leading researchers, and the practical application of modern regenerative medicine technologies. We will embark on a detailed exploration of Platelet-Rich Plasma (PRP) and a groundbreaking adjunct, Protein Concentrate, derived from Platelet-Poor Plasma (PPP). My commitment is to transparency and precision, ensuring that for every patient, we understand the exact composition of the injectate we are using. We will move beyond the outdated metric of “x-fold concentration” and delve into the critical importance of absolute platelet dose, platelet recovery rates, and the specific cellular components of our biologics, including reductions in pro-inflammatory cells such as granulocytes and Red Blood Cells (RBCs).
This discussion will thoroughly unpack the science behind a specific PRP preparation system I use in my clinic, which boasts an impressive average platelet recovery rate of 83% and can yield a platelet dose of approximately 10.8 billion from a single 60 cc blood draw. We’ll examine real-world hematocrit data from my practice to illustrate these concepts, demonstrating how we can achieve a 94% recovery rate in a given sample, thereby ensuring a potent, purified final product. A significant portion of our journey will be dedicated to a topic often overlooked: the untapped potential of Platelet-Poor Plasma (PPP). Historically discarded, PPP is now understood to be a rich reservoir of crucial proteins and growth factors. We will detail the process for transforming PPP into a potent Protein Concentrate using a specialized fluid-reduction filter. This process concentrates vital molecules such as Alpha-2-Macroglobulin (A2M), Interleukin-1 Receptor Antagonist (IL-1ra), and various growth factors, creating a powerful therapeutic agent for the management of conditions such as osteoarthritis. We will explore the physiological mechanisms of these molecules, explaining how A2M acts as a “trap” for catabolic enzymes that degrade cartilage and how IL-1ra blocks inflammatory pathways, thereby reducing pain and inflammation.
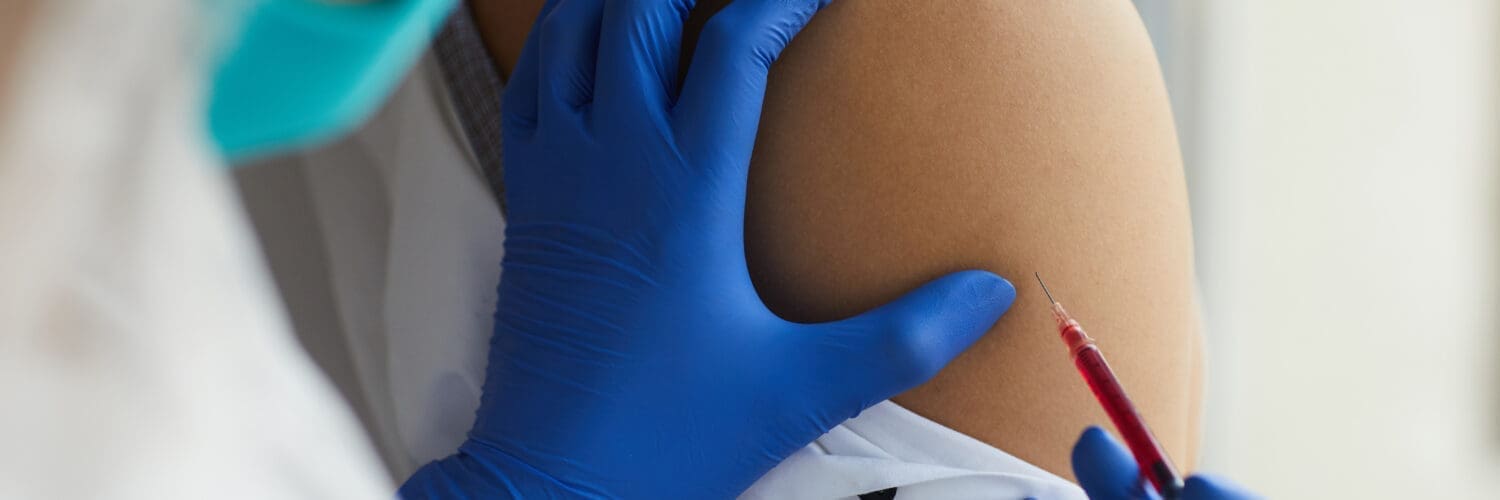
Furthermore, we will transition from the laboratory to the clinic, discussing the patient journey in its entirety. This includes the meticulous pre-procedure consultation, which, in my experience, requires a thorough 36-minute discussion to cover the diagnosis, all treatment options, and financial considerations. We’ll cover crucial pre-procedure patient optimization strategies, from managing medications like anti-inflammatories to the importance of hydration and nutritional support. We’ll then walk through a specific clinical application: an ultrasound-guided acromioclavicular (AC) joint injection. I will describe my preferred out-of-plane technique, patient positioning, and the rationale behind my systematic approach to treating comprehensive shoulder pathology. The post-procedure phase is equally critical. We will discuss tailored rehabilitation protocols, the adjunctive use of laser therapy (photobiomodulation) to accelerate healing and manage post-injection discomfort, and the nuanced debate surrounding the use of ice versus heat. This comprehensive guide aims to provide fellow practitioners and informed patients with a deep, evidence-based understanding of how to harness the body’s own healing potential with precision, efficacy, and a patient-centered focus.
Maximizing Therapeutic Potential: The Science of High-Quality PRP Preparation
In my practice, the pursuit of excellence in regenerative medicine begins with a fundamental principle: quality. When we talk about Platelet-Rich Plasma (PRP), the quality of the final product is paramount. It’s not enough to draw blood and spin it in a centrifuge. We must be meticulous, scientific, and data-driven in our approach. My choice of a preparation system is based on several key pillars that ensure I am delivering the most effective and predictable treatment to my patients. These pillars are quality, versatility, reproducibility, and output.
The Critical Importance of High Platelet Recovery
The first and most critical quality metric is elevated platelet recovery. The goal of PRP preparation is to capture as many platelets as possible from the initial whole-blood draw and concentrate them into a smaller therapeutic volume. Many systems on the market lose a significant percentage of platelets during processing. These lost platelets end up either in the discarded plasma layer (the platelet-poor plasma or serum) or remain trapped within the red blood cell (RBC) stack at the bottom of the tube.
In my clinic, we utilize a system that achieves an average platelet recovery of about 83%. This is a clinically significant figure. It means that for every 100 platelets drawn from the patient, 83 are successfully captured and included in the final injectate. This high recovery rate is the foundation of a potent therapeutic dose. To illustrate this with data from my own practice, let’s consider a recent case.
We started with a 60 cc whole-blood draw. A precentrifugation analysis using a hematocrit analyzer showed the patient’s baseline platelet count to be 265,000 per microliter (265 × 10³/µL). To understand the total number of platelets we started with, we perform a simple calculation:
- 265,000 platelets/µL x 60,000 µL (60cc) = 15.9 billion total platelets in the initial sample.
This 15.9 billion represents the total “option” of platelets available for us to harvest. After a single, efficient 10-minute centrifugation, we produced 7 cc of PRP. We then analyzed this final product. The platelet measurement in the PRP was an astounding 2,128,000 per microliter (or 2,128 x 10³/µL).
To calculate the final platelet dose delivered to the patient, we multiply this new concentration by the final volume:
- 2,128,000 platelets/µL x 7,000 µL (7cc) = 14.89 billion total platelets in the final 7cc injectate.
By comparing the platelets in the final product to the platelets in the initial draw, we can determine our recovery rate for this specific patient:
- (14.89 billion / 15.9 billion) x 100 = 93.6% or a 94% recovery rate.
This is a testament to the efficiency of the system. We successfully captured nearly all of the available platelets, creating an incredibly rich and potent biologic.
Moving Beyond X-Fold: The Importance of Absolute Platelet Dose
For years, the industry standard for describing PRP concentration was the “x-fold” metric—for example, a “5x” or “7x” concentration over baseline. While this provided a rough estimate, it is imprecise and often misleading. The final therapeutic effect is determined not by a relative increase but by the absolute number of platelets injected into the target tissue.
In the example above, the final concentration (2,128) is roughly 8 times the baseline concentration (265). However, stating it’s an “8x” PRP is less informative than stating that the patient received a 14.89 billion-unit dose. The absolute dose is the number that truly matters for stimulating a healing response. The system I use consistently averages about 10.8 billion platelets from a single 60 cc blood draw, which corresponds to roughly a 10x increase in concentration from the average baseline. This provides a powerful, therapeutically relevant dose for a wide range of orthopedic conditions.
Purity of the Injectate: Minimizing Pro-Inflammatory Cells
The quality of PRP is not just about what we concentrate on, but also about what we remove. The ideal PRP should be rich in platelets and their associated growth factors but poor in cells that can cause a counterproductive inflammatory flare. Specifically, we want to minimize the presence of granulocytes (a type of white blood cell) and Red Blood Cells (RBCs).
- Granulocytes: While some white blood cells are beneficial, high concentrations of granulocytes, particularly neutrophils, can release pro-inflammatory cytokines and proteolytic enzymes that may exacerbate pain and potentially damage tissue in the short term. Looking back at our sample data, the granulocyte count was significantly reduced relative to the whole-blood baseline, resulting in a less inflammatory injectate.
- Red Blood Cells (RBCs): RBCs are highly inflammatory when introduced into a synovial joint or soft tissue. Their lysis (breakdown) can cause significant pain, swelling, and oxidative stress. A “clean” or “amber” PRP is visually indicative of low RBC contamination. Our analysis confirms this. The hematocrit data for the final PMP (Platelet-Rich Plasma) sample indicated an RBC concentration below 0.1. This is a virtually pure preparation, minimizing the risk of a painful post-injection flare and allowing the regenerative components to work in a more favorable microenvironment.
As a clinician, having access to this level of data is non-negotiable. I perform these analyses in my clinic for every patient. I need to know the precise dose, the white blood cell contribution, and the RBC contribution. This allows me to confidently dial in the exact formulation for each unique patient and their specific pathology.
The Power of Versatility and Reproducibility
Another cornerstone of my approach is versatility. The system I employ allows me to precisely control not only the final volume of the injectate but also its composition. By carefully taking platelets from both the buffy coat (the thin layer containing platelets and white blood cells) and the adjacent RBC stack, I have fine-tuned control over the final product. This flexibility is crucial. A tendon injection might require a smaller, highly concentrated volume, while a large joint like the knee might benefit from a larger volume to adequately bathe the articular surfaces. This system provides both simplicity and reproducibility in its standard protocol, as well as expert control to create a flexible volume and a flexible injectate tailored to clinical needs.
Unlocking the Hidden Potential of Platelet-Poor Plasma (PPP)
For many years in the field of regenerative medicine, the clear, yellowish fluid left over after PRP separation—the Platelet-Poor Plasma (PPP)—was discarded. It was considered the “waste product” of the procedure. However, modern, evidence-based research has illuminated a profound truth: PPP is not waste at all. It is a treasure trove of biologically active proteins and molecules that play crucial roles in healing, modulation of inflammation, and tissue homeostasis. Instead of discarding this “liquid gold,” we can now concentrate it to create a powerful therapeutic adjunct called Protein Concentrate.
The Rich Composition of Platelet-Poor Plasma
Before we discuss the concentration process, it’s vital to understand what PPP contains. While it is “poor” in platelets relative to the PRP fraction, it is by no means devoid of them. Our analysis shows that we can scavenge a significant number of additional platelets from this fraction. More importantly, PPP is rich in soluble proteins critical for joint health and tissue repair. These include:
- Alpha-2-Macroglobulin (A2M): A very large and powerful protease inhibitor.
- Hepatocyte Growth Factor (HGF): A potent growth factor involved in tissue regeneration.
- Soluble TNF-? Receptors (sTNF-R): Molecules that can bind and neutralize the pro-inflammatory cytokine TNF-?.
- Platelet-Derived Growth Factor (PDGF-BB): A key growth factor for cell proliferation and angiogenesis.
- Interleukin-1 Receptor Antagonist (IL-1ra): A naturally occurring anti-inflammatory protein that blocks the action of the highly catabolic cytokine, Interleukin-1.
- Epidermal Growth Factor (EGF): Important for cell growth and differentiation.
- Water: The primary component, which we will strategically remove.
In its native state, the concentrations of these beneficial proteins are relatively dilute. The key is to concentrate them, amplifying their therapeutic power.
The Concentration Process: Creating Protein Concentrate
The transformation of PPP into Protein Concentrate is achieved through a sophisticated yet elegant process using a specialized fluid-reduction filter. Here’s a step-by-step breakdown of how we accomplish this in the clinic:
- Preparation: The process begins with our harvested PPP, which was set aside during the initial PRP preparation. We use a sterile, closed system to ensure no contamination. The core of this system is the fluid volume reducer, which contains a pre-wetted 15-kilodalton filter. This kilodalton size is a critical detail; it refers to the molecular weight cutoff of the filter’s pores. The pores are small enough to allow water molecules to pass through but large enough to retain the much larger, therapeutically important proteins, such as A2M (720 kDa), IL-1ra, and various growth factors.
- Priming the System: We connect the syringe containing the PPP to the filter system. The first step is to gently push a small amount of the PPP through the filter to prime it and expel any excess air, ensuring a smooth and efficient filtration process.
- Creating the Vacuum: An empty 60 mL vacuum-lock syringe is attached to the filter’s water-waste port. By pulling back the plunger and locking it, we create a sustained vacuum. This vacuum acts as the driving force for the dehydration process.
- Dehydration and Concentration: The PPP is then pushed back and forth between two syringes, flowing across the filter membrane. As it passes over the filter, the vacuum on the other side pulls the water out of the plasma and into the waste syringe. The valuable proteins and remaining platelets, being too large to pass through the 15 kDa pores, are retained and become progressively more concentrated in the diminishing volume of plasma.
- Achieving Target Volume: Our clinical goal is to dehydrate the sample by about 75%. For example, if we start with 20 cc of PPP, we aim to reduce it to approximately 5 cc of Protein Concentrate. This back-and-forth process continues until all the desired water has been removed and the target volume is achieved.
- Harvesting the “Liquid Gold”: Once the concentration is complete, the stopcock is repositioned, the waste syringe is removed, and the final, highly concentrated product is drawn into a sterile syringe. What we are left with is a viscous, protein-rich solution—the Protein Concentrate.
The Therapeutic Power of Concentrated Proteins
This process of concentrating PPP results in a dramatic increase in the bioavailability of its key components. We see a fivefold increase in Alpha-2-Macroglobulin (A2M) concentration compared to baseline, and a twofold increase in soluble TNF-? receptors. Most impressively, we achieve an IL-1ra-to-IL-1 ratio. ratio of 100:1, which is of profound clinical significance in combating inflammation.
Furthermore, this process allows us to scavenge an additional 1.6 billion platelets on average that would otherwise have been discarded. These platelets are added back to the primary PRP injectate or used in combination to boost the overall therapeutic dose further.
The Physiological Rationale: Why Protein Concentrate is a Game-Changer for Osteoarthritis
Historically, physicians focused solely on the PRP fraction and discarded the PPP. Current research and my own clinical observations strongly support the notion that, for conditions like osteoarthritis (OA), the components of the PPP are just as important, if not more so, in modulating the disease process.
The Role of Alpha-2-Macroglobulin (A2M)
Osteoarthritis is characterized by a chronic, low-grade inflammatory state within the joint. This inflammation leads to the upregulation of catabolic enzymes—proteases such as matrix metalloproteinases (MMPs) and ADAMTS—that degrade articular cartilage. This creates a destructive feedback loop where cartilage breakdown products fuel further inflammation.
Alpha-2-Macroglobulin (A2M) is one of the body’s most powerful defenses against these destructive enzymes. It functions as a “trap.” When a catabolic enzyme contacts A2M, the A2M molecule undergoes a conformational change, closing around the enzyme and irreversibly binding it. This A2M-enzyme complex is then cleared from the joint by macrophages, effectively removing the “cartilage-eating” enzymes from the synovial fluid.
The problem in an arthritic joint is twofold. First, the chronic production of catabolic enzymes can overwhelm the joint’s natural supply of A2M. Second, A2M is a very large molecule, weighing in at 720 kilodaltons. This immense size prevents it from readily migrating from the bloodstream into the joint’s synovial fluid. Therefore, the joint is often deficient in this crucial protective protein.
By injecting a concentrated dose of A2M directly into the joint, we are delivering a powerful intervention. We are re-arming the joint with the tools it needs to counteract the degradative process, halt cartilage breakdown, and break the negative feedback loop of chronic inflammation.
The Power of Interleukin-1 Receptor Antagonist (IL-1ra)
Another key villain in the story of osteoarthritis is Interleukin-1 (IL-1). This cytokine is a master regulator of inflammation and pain. When IL-1 binds to its receptors on chondrocytes (cartilage cells) and synoviocytes (cells of the joint lining), it triggers a cascade of events that leads to increased production of catabolic enzymes, inhibition of new cartilage matrix synthesis, and the generation of pain signals.
The body has a natural counterbalance to IL-1: a protein called Interleukin-1 Receptor Antagonist (IL-1ra). As its name suggests, IL-1ra acts as a competitive inhibitor. It has a similar shape to IL-1 and can bind to the same IL-1 receptors on the cell surface. However, when IL-1ra binds, it does not activate the receptor. It simply occupies the space, physically preventing IL-1 from binding and initiating its inflammatory cascade.
In an arthritic joint, the balance is skewed. There is far more pro-inflammatory IL-1 than anti-inflammatory IL-1ra. By creating Protein Concentrate, we can deliver a solution with a 100:1 IL-1ra-to-IL-1? ratio. When this is injected into the joint, we flood the environment with this protective antagonist protein. The IL-1ra molecules saturate the available receptors, effectively silencing the inflammatory signals being sent by IL-1. The clinical result is a significant reduction in both pain and inflammation, creating a more favorable environment for PRP growth factors to initiate repair.
By combining high-dose, purified PRP with this potent, anti-inflammatory, and anti-catabolic Protein Concentrate, we are not just stimulating repair; we are fundamentally changing the diseased microenvironment of the arthritic joint. We are addressing both sides of the equation: promoting regeneration while simultaneously halting degradation and inflammation. This comprehensive, dual-pronged approach represents a significant advancement in the non-operative management of osteoarthritis.
5 Things You Need to Know About Ligamentous Injuries Before They Get Worse- Video
The Comprehensive Patient Journey: From Consultation to Recovery
A successful orthobiologic outcome is not solely dependent on the quality of the injectate or the precision of the injection. It is the culmination of a meticulous, patient-centered process that begins long before the needle ever touches the skin. In my practice, this journey is divided into three critical phases: the pre-procedure discussion and optimization, the procedure itself, and the post-procedure care and rehabilitation.
The Pre-Procedure Discussion: Setting the Foundation for Success
I am often asked how I manage my clinic flow, and I am steadfast in my response: my initial orthobiologics consultation takes 36 minutes. I have tried to condense it, but I have found that this is the minimum time required to be truly thorough and to honor the commitment my patients are making. This consultation is not just an evaluation; it’s a collaborative partnership.
- Arriving at the Right Diagnosis: The first step is to achieve diagnostic certainty. This involves a comprehensive history, a detailed physical examination, and, crucially, the use of in-office diagnostic ultrasound and a review of other imaging, such as MRI or X-ray. We must pinpoint the exact anatomical pain generators. Is the patient’s shoulder pain coming from the glenohumeral joint, the AC joint, the supraspinatus tendon, or a combination of these? A vague diagnosis leads to a vague and often ineffective treatment plan.
- Discussing All Options: My role as a clinician is to serve as an educator and guide. I discuss the full spectrum of treatment options available to the patient, from conservative measures like physical therapy and activity modification to interventional options like steroid injections, orthobiologics, and surgery. We discuss the risks, benefits, and expected outcomes of each. The patient must understand where regenerative medicine fits into this continuum.
- Recommending the Most Viable Option: Based on the diagnosis, the severity of the condition, the patient’s goals, and the current scientific evidence, I then recommend what I believe is the most viable option. For many musculoskeletal conditions, this is an orthobiologic procedure. This is where we discuss the specifics of PRP, Protein Concentrate, or other cellular therapies, personalizing the recommendation to their unique pathology.
- Managing Medications and Expectations: This is a critical part of the pre-procedure discussion.
- Corticosteroid Injections: We need to know if and when the patient last had a steroid injection. Corticosteroids can be toxic to local cells and can suppress the very healing response we are trying to stimulate. A washout period of several weeks to months is often required before proceeding with a regenerative therapy.
- Anti-inflammatories (NSAIDs): Standard over-the-counter NSAIDs like ibuprofen (Advil) and naproxen (Aleve) interfere with platelet function by inhibiting the COX-1 pathway, which is essential for the initial inflammatory phase of healing. I instruct patients to discontinue these medications. If a patient requires an anti-inflammatory for another medical condition and cannot stop, I will often transition them to meloxicam. Some newer studies suggest that meloxicam, being more COX-2 selective, may be more compatible with orthobiologic treatments than traditional NSAIDs. If they can come off NSAIDs entirely, we may recommend supplements that can help manage pain and inflammation without disrupting platelet function, such as curcumin or boswellia.
- Hydration: Proper hydration is essential for a good blood draw and optimal plasma volume. I instruct my patients to begin consciously hydrating with plenty of water two days before their scheduled procedure.
- Diet: While the evidence is still emerging and somewhat controversial, there has been some discussion that intermittent fasting before the blood draw may improve PRP quality. At a minimum, I ask patients to have a healthy, light breakfast and avoid a heavy, fatty meal, which can lead to lipemic (fatty) plasma that can interfere with processing.
- The Financial Conversation: Transparency is key. The vast majority of orthobiologic procedures are not reimbursed by insurance. It is my ethical responsibility to have an open and honest conversation with the patient about the cost of the procedure before they commit. There should be no surprises. My Medicare patients are required to sign an Advanced Beneficiary Notice (ABN) before any cash-pay procedure, acknowledging that they understand Medicare will not cover the cost and that they are financially responsible.
Pre-Procedure Optimization: Stacking the Deck in the Patient’s Favor
Beyond the immediate pre-procedure instructions, I believe in optimizing the patient’s systemic environment to enhance their cellular health and metabolic function. This often involves pre-procedure blood work to identify any underlying nutritional deficiencies or metabolic issues. Based on these results, I may recommend a targeted supplementation protocol to improve mitochondrial function, reduce systemic inflammation, and ensure their cells have all the necessary building blocks for repair. My philosophy is simple: I want to stack the deck in my patient’s favor in every conceivable way to maximize their potential for a successful outcome.
Clinical Application: Ultrasound-Guided Acromioclavicular (AC) Joint Injection
Let’s transition from theory to practice and walk through a common procedure I perform in my clinic: an injection of the acromioclavicular (AC) joint. This small joint, where the clavicle (collarbone) meets the acromion of the scapula (shoulder blade), is a frequent source of pain, often due to arthritis or injury. Precise, image-guided delivery of the biologic is essential for success.
Patient Positioning and Technique
For most shoulder procedures, including those involving the AC joint, I have the patient seated and upright. This position provides excellent access to the various structures of the shoulder and, from a workflow perspective, allows me to efficiently move from one injection site to another when performing a comprehensive shoulder treatment.
- A Note on Vasovagal Risk: Any needle-based procedure carries a risk of a vasovagal response (fainting). I always remain vigilant and am prepared to quickly convert the patient to a lateral decubitus (lying on their side) position if they begin to feel lightheaded or dizzy.
My preferred technique for an AC joint injection is an out-of-plane approach. This means the needle enters the skin perpendicular to the long axis of the ultrasound probe.
- Site Identification and Preparation: I begin by palpating the joint, then place the ultrasound probe in a coronal orientation directly over it. The goal is to get a clear image of the distal clavicle on one side and the acromion on the other, with the dark, hypoechoic joint space in between. I use a skin marker to mark my intended entry point. The area is then meticulously prepped using a sterile technique, typically with Betadine followed by alcohol, to minimize any risk of infection. The skin is then anesthetized with a small amount of local anesthetic, such as lidocaine.
- The Injection: An Out-of-Plane, Posterior-to-Anterior Approach: I place the ultrasound probe on the anterior aspect of the AC joint, keeping the joint space centered on the screen. I prefer to “hide” the injection from the patient’s view by entering the joint from its posterior aspect. My target is the V-shaped capsule of the joint, which is typically about 5 centimeters deep beneath the skin.
- Visualizing the Needle: With an out-of-plane technique, the needle shaft is not seen in its entirety. Instead, as the needle tip passes through the ultrasound beam, it appears as a bright white, hyperechoic dot. My goal is to triangulate and guide this bright dot directly into the dark joint space.
- Needle Placement and Injection: As I advance the needle, I watch the ultrasound screen intently. On the first pass, I might find my needle tip is “saddled up next to the bone”—too lateral or too medial. This requires a slight redirection. I’ll pull back slightly and adjust my angle to a more central position, aiming for the heart of the joint. Once I see the bright white dot of the needle tip perfectly positioned within the joint space, I inject the PRP. On the ultrasound, you can see the anechoic (black) flow of the PRP as it fills and distends the joint capsule.
- Volume Considerations: The AC joint is a small space. It typically holds between 1.1 and 6 cc of fluid. Over-distending the joint can be painful, so it’s important to inject slowly and stop when appropriate resistance is felt. This is where the versatility of the PRP system is key, allowing me to prepare a small, precise volume for a small joint like this.
A Comprehensive Shoulder Treatment Workflow
The seated position is particularly efficient when I am treating multiple pathologies in the same shoulder, which is often the case. My typical workflow, after administering an interscalene nerve block for patient comfort, is to work from posterior to anterior:
- Posterior Glenohumeral Joint Injection: Start with the main shoulder joint from a posterior approach.
- AC Joint Injection: Come up over the top of the shoulder to perform the out-of-plane AC joint injection as described above.
- Rotator Cuff and Biceps Tendon: Move to the front and side of the shoulder to address the rotator cuff tendons (infraspinatus, supraspinatus, subscapularis) and the biceps tendon in both its long and short axes.
This systematic back-to-front approach allows me to work efficiently while the nerve block is taking full effect, maximizing patient comfort and clinical productivity. Using a small 25-gauge needle for the AC joint injection also helps to minimize patient discomfort during the procedure.
Post-Procedure Care: Guiding the Healing Process
The procedure itself is just the beginning of the regenerative cascade. The post-procedure phase is where we guide the patient’s body through the healing process and manage their expectations. A structured and well-communicated post-procedure plan is critical for optimizing outcomes and ensuring patient compliance.
Immediate Post-Procedure Management
For a comprehensive shoulder procedure, I typically place the patient in a sling for one to two days. This serves several purposes. It provides comfort, limits excessive early movement that could disrupt the newly placed biologic, and serves as a visual advertisement to the patient and those around them that something significant has been done, encouraging them to protect the shoulder.
The Role of Rehabilitation
Rehabilitation is not optional; it is a mandatory component of a successful orthobiologic treatment. The biologic provides the “seed and fertilizer,” but the mechanical loading and progressive stress provided by physical therapy are the signals that tell the cells what to do and how to align.
- Tailoring Rehab Protocols: I have found a very effective and efficient way to create rehab protocols, particularly for intratendinous work like a rotator cuff injection. I leverage the excellent, evidence-based post-surgical protocols developed by orthopedic surgeons. For a non-surgical regenerative procedure, I will take the corresponding post-surgical rehab protocol and instruct the patient and their physical therapist to begin at week six of that protocol. For example, after a rotator cuff PRP injection, the patient would start a post-surgical rotator cuff repair protocol at the six-week mark. This bypasses the initial, highly restrictive immobilization phase required for a surgical repair and starts them right at the point of gentle, progressive strengthening and range of motion.
Managing Post-Injection Inflammation and Pain
It is crucial to educate the patient that an inflammatory response is a necessary and desirable part of the healing process. The goal is not to eliminate it but to manage it and guide it.
- Discontinuation of Anti-inflammatories: I instruct patients to continue to avoid NSAIDs for at least two weeks post-procedure, and longer if possible, to allow the natural inflammatory cascade to unfold.
- Safety with Blood Thinners: From a safety perspective, we provide clear instructions on when to restart any blood thinners, balancing the risk of bleeding with the patient’s underlying medical need for anticoagulation. This is always done in consultation with their prescribing physician.
The Ice vs. Heat Debate and the Power of Photobiomodulation
Managing post-injection pain is a key concern. My approach is to use modalities that work with the body’s biology, not against it.
- Laser Therapy (Photobiomodulation): I am a strong advocate for using laser therapy, starting as early as the day of or the day after the procedure. The mechanism of photobiomodulation involves the absorption of specific wavelengths of light (typically in the red and near-infrared spectrum) by mitochondria. This enhances ATP production, the cell’s energy currency, thereby accelerating cellular metabolism and the healing process. Laser therapy also helps manage pain by promoting nitric oxide release, which improves local circulation, and by modulating nerve conduction. By using a laser, we can accelerate the inflammatory curve, shortening the duration of the most uncomfortable phase for the patient.
- Heat vs. Ice: I encourage using near-infrared (NIR) light therapy or moist heat. These modalities increase blood flow, relax tissues, and support cellular metabolism. I generally advise patients to avoid ice if possible. The traditional RICE (Rest, Ice, Compression, Elevation) protocol was designed for acute traumatic injuries to limit swelling. In the context of a regenerative procedure, where we are trying to stimulate a biological response, cold therapy can be counterproductive. Ice causes vasoconstriction, reduces local blood flow, and slows cellular metabolism, potentially halting the release of growth factors and blunting the very response we are trying to create. However, I am also a pragmatist. If a patient’s post-injection pain is severe and ice is the only modality they have access to that provides relief, I will allow it, but it is not my first choice. My preference is always laser, NIR, or heat.
By providing a clear, structured post-procedure plan, we empower the patient to be an active participant in their own recovery, leading to better compliance, managed expectations, and ultimately, superior clinical outcomes.
Engaging with the Orthobiologics Community
I am deeply committed to education and collaboration within the medical community. To that end, I want to share some resources and upcoming opportunities for colleagues interested in expanding their knowledge and skills in this exciting field.
Resource Library: For Apex Biologics customers, you have access to an extensive resource library containing over 130 research articles, procedural videos (including the Protein Concentrate processing video), FAQs, and specialty-specific educational webinars. This is an invaluable tool for staying current with the evidence. If you are not a current customer, you can access the site by visiting apexbiologics.com.
Orthobiologic Readiness Assessment: For clinicians contemplating integrating regenerative medicine into their practice, it can be daunting to know where to start. We have developed a short quiz to help you assess your readiness for orthobiologics. It evaluates factors like clinical competence, workflow readiness, and capital considerations to provide you with a personalized pathway to integration, whether you’re looking for a lean startup or a full practice launch.
Upcoming Educational Events:
I believe strongly in the power of hands-on learning and peer-to-peer collaboration. I am excited to be hosting and participating in several upcoming events:
- Hands-On Workshop in Clearwater, FL (March 22-23, 2024): This complimentary workshop offers business-focused training on patient-centric branding, effective pricing strategies, and the use of AI to expand your practice. The second day is dedicated to hands-on cadaver training for microfragmented adipose (M-Fat) processing, bone marrow aspirate (BMA) harvest techniques, and advanced PRP and Protein Concentrate preparation.
- True MD Institute Event in Arizona (May 17-19, 2024): Alongside my colleagues Atima D’Souza and Colin Rigney, I am hosting an exclusive three-day live event at the luxurious Phoenician resort. This event is designed to help a limited group of 40 physicians engineer a predictable, cash-pay regenerative medicine practice. It includes an online MSK ultrasound primer, AI implementation blueprints, and personalized business strategy sessions.
- Webinar: Regenerative Solutions for the Hip (March 12, 2024): My colleague, Dr. Baudouin, a board-certified pain medicine and anesthesiology physician, will be presenting a webinar on differentiating hip pathology and exploring the evidence for PRP in treating hip osteoarthritis and gluteal tendinopathy.
- Webinar: Intrameniscal Injections (March 19, 2024): I will be back in two weeks to lead a deep-dive discussion on the techniques and rationale for performing not just intra-articular knee injections, but technically demanding intrameniscal and meniscocapsular junction injections.
Continuous learning is the lifeblood of medical advancement, and I encourage all my colleagues to engage with these opportunities.
Clinical Q&A: Addressing Common Concerns
During these educational sessions, I always welcome questions from my colleagues. The ensuing discussions often highlight important clinical nuances and practical challenges. Here are a few insightful questions from a recent session.
Question 1: Photobiomodulation, Cellular Therapies, and Tumorigenesis
Question: “For M-Fat (microfragmented adipose) and BMAC (bone marrow aspirate concentrate) procedures, are there any concerns that starting photobiomodulation (laser therapy) one week later could cause the cells, especially the mesenchymal stem cells in M-Fat, to turn into tumors? I saw a theoretical risk mentioned by an AI chatbot.”
My Response: This is an excellent and important question that touches on the safety of our modalities. My answer is definitive: there is no evidence to support this concern.
First, let’s look at the therapies themselves. Large-scale case series and clinical studies on both M-Fat and BMAC have been conducted for years. The data consistently show no significant increase in tumorigenesis (tumor formation) associated with the use of these autologous (from the patient’s own body) cellular therapies in orthopedic applications. The cells are being placed in an orthopedic environment and respond to the local cues to differentiate into musculoskeletal tissues or, more commonly, to act via paracrine signaling to orchestrate a healing and anti-inflammatory response.
Second, let’s clarify the mechanism of photobiomodulation (PBM). PBM does not work by causing uncontrolled cellular proliferation. Its primary mechanism is the absorption of photons by cytochrome c oxidase in mitochondria, which enhances electron transport chain efficiency and increases ATP availability. It’s like giving a factory more power to do its intended job more efficiently; it doesn’t change what the factory is designed to produce. We are not using high-power, ablative, or ionizing lasers that cause genetic damage. This is low-level laser therapy designed to enhance normal cellular metabolism.
Additionally, PBM has other beneficial mechanisms for pain relief, including enhanced nitric oxide release, which acts as a vasodilator and improves local circulation, and modulation of inflammatory pathways.
In my practice, I don’t even wait a week. I often start PBM the day of or the day after the procedure. I find it an excellent tool for managing post-injection pain and inflammation, and it helps “jump-start” the metabolic activity of the cells we’ve just injected. The theoretical concern raised by a general AI chatbot highlights the importance of relying on peer-reviewed scientific literature and experienced clinical judgment rather than unverified sources. In this case, both the clinical data and the known biological mechanisms confirm the safety of combining these therapies.
Question 2: Post-Meniscectomy Knee Pain and Treatment Strategy
Question: “I have a patient who had two arthroscopic lateral meniscus repairs years ago and is now presenting with pain. I know arthroscopic surgery can lead to early-onset OA. When patients come back years later, is the pain more likely related to the meniscus or the OA that was kick-started by the procedure?”
My Response: This question requires us to be very precise with our language and our understanding of the pathology. The premise contains a few points that need clarification.
First and foremost, we must differentiate between two very different surgical procedures: arthroscopic meniscal repair and arthroscopic partial meniscectomy.
- A meniscal repair is a procedure where the surgeon sutures the torn meniscus back together, preserving the tissue.
- A partial meniscectomy is a procedure where the surgeon trims out and removes the torn portion of the meniscus.
Upon further clarification, the practitioner confirmed the patient actually had a partial lateral meniscectomy (a “trimming”) about 12 years prior, not a repair. This is a critical distinction.
The statement that “arthroscopic surgery kicks off arthritis” is not accurate. Arthroscopic surgery itself does not accelerate arthritis. In fact, a simple fluid lavage (washing out the joint) can sometimes provide temporary relief from arthritic symptoms. However, the consequences of what is done during that arthroscopy are what matter.
- A successful meniscal repair that leads to healing is chondroprotective. By restoring the structural integrity of the meniscus, we restore its function as a shock absorber, which protects the articular cartilage from excessive wear and tear.
- A partial meniscectomy, on the other hand, does the opposite. When we cut out a piece of the meniscus, especially the lateral meniscus, we permanently remove a critical shock absorber. This leads to a dramatic increase in contact pressures and point loading on the articular cartilage in that knee compartment. This is what accelerates the arthritic process.
So, to answer the question directly: Is it expected that 12 years after a lateral partial meniscectomy, a patient would have accelerated arthritic change? Absolutely. We know this to be true from extensive research. The surgery didn’t cause the arthritis; the removal of the meniscus led to biomechanical changes that caused it. In fact, the current literature does not support performing a partial meniscectomy in a knee with significant arthritic changes, as outcomes are often poor.
Now, regarding the treatment strategy for this patient:
The primary pathology is now an arthritic knee, secondary to the meniscectomy. Therefore, the treatment should be directed at the arthritis. The first step is a thorough diagnostic workup. I would use ultrasound to assess:
- The severity of the arthritis (joint space narrowing, osteophytes).
- The presence of a joint effusion (swelling).
- The status of the remaining meniscus (is it diminutive, or has a new tear developed?).
- The presence of any bone marrow lesions (which may require a more advanced cellular therapy like BMAC).
The choice of injectate would then be based on the disease state.
- For mild to moderate arthritis, a high-dose PRP combined with Protein Concentrate would be an excellent choice to modulate inflammation and provide growth factors.
- For severe or end-stage arthritis, especially with bone marrow lesions, a more robust cellular therapy, such as bone marrow concentrate (BMAC) or adipose-derived stem cells, might be indicated.
Would I inject into the meniscus itself? If the meniscus is simply diminutive (small) from the previous resection and there is no new, distinct tear, then an intrameniscal injection is not indicated. The pathology is not in the remaining meniscal tissue; the pathology is the arthritis in the joint. The treatment should be an intra-articular (into the joint space) injection to treat the entire joint environment.
This case perfectly illustrates why a nuanced and precise diagnosis is the absolute prerequisite for developing a personalized and effective treatment plan. We must treat the pathology that exists today, not just the history of what was done 12 years ago.
Summary
This educational post provides a comprehensive overview of advanced techniques and clinical reasoning in orthobiologics. We began by establishing the critical importance of producing a high-quality Platelet-Rich Plasma (PRP), moving beyond the outdated “x-fold” metric to focus on the clinically relevant absolute platelet dose and high platelet recovery rates. Using real-world data from my clinic, we demonstrated how a superior system can achieve over 90% platelet recovery, yielding a potent therapeutic dose while simultaneously purifying the injectate by drastically reducing pro-inflammatory granulocytes and Red Blood Cells (RBCs). A significant focus was placed on the paradigm shift in valuing Platelet-Poor Plasma (PPP). We detailed the process of transforming this once-discarded byproduct into a powerful Protein Concentrate rich in anti-inflammatory and anti-catabolic molecules. We explored the physiological mechanisms of key proteins like Alpha-2-Macroglobulin (A2M), which neutralizes cartilage-degrading enzymes, and Interleukin-1 Receptor Antagonist (IL-1ra), which blocks inflammatory pathways, explaining why this concentrate is a game-changer for conditions like osteoarthritis.
The discussion then bridged the gap from the lab to the clinic, outlining the comprehensive patient journey. We emphasized the necessity of a thorough pre-procedure consultation for diagnosis, education, and patient optimization, including medication management and nutritional support. We provided a practical, step-by-step guide to performing an ultrasound-guided acromioclavicular (AC) joint injection, detailing my out-of-plane, posterior-to-anterior technique. We also covered the vital post-procedure phase, discussing tailored rehabilitation protocols, the adjunctive use of laser therapy (photobiomodulation) to accelerate healing, and the nuanced rationale for preferring heat-based modalities over ice. Finally, we addressed common clinical questions, clarifying the safety of combining cellular therapies with laser therapy and outlining the appropriate diagnostic and treatment strategies for a patient with post-meniscectomy knee pain.
Conclusion
The field of orthobiologics is evolving rapidly, demanding that we, as clinicians, remain lifelong learners, constantly refining our techniques and deepening our understanding of the underlying physiology. The principles outlined here—a commitment to data-driven quality, a focus on the absolute platelet dose, the strategic use of all blood components, including Protein Concentrate, and a comprehensive, patient-centric approach from consultation through rehabilitation—represent the cornerstones of a modern, effective regenerative medicine practice. By harnessing the power of A2M to protect cartilage and IL-1ra to quell inflammation, in concert with the regenerative signaling of high-dose PRP, we can offer our patients a sophisticated, multifaceted intervention that addresses the complex pathology of musculoskeletal disease. The ultimate goal is to move beyond simply managing symptoms and instead fundamentally alter the disease microenvironment, empowering the body’s innate capacity for healing to achieve durable, meaningful, and life-changing outcomes for our patients.
Key Insights
- Dose is King: The most important metric for PRP effectiveness is the absolute platelet dose, not the “x-fold” concentration. A higher dose delivers more growth factors to the target tissue.
- Purity Matters: A high-quality PRP is not only rich in platelets but also low in pro-inflammatory cells such as granulocytes and RBCs, leading to better outcomes and less post-injection pain.
- Don’t Waste the Plasma: Platelet-Poor Plasma (PPP) is a valuable source of anti-inflammatory and cartilage-protective proteins. Concentrating it into Protein Concentrate to harvest A2M and IL-1ra provides a powerful tool against the degenerative and inflammatory aspects of osteoarthritis.
- Precision is Paramount: Ultrasound guidance is the standard of care. It ensures the accurate delivery of the biologic to the precise anatomical target, transforming a “blind” procedure into a precise, visually confirmed intervention.
- Healing is a Process, Not an Event: A successful outcome depends on a comprehensive journey that includes meticulous pre-procedure optimization, a precise procedure, and a structured post-procedure rehabilitation plan. Modalities like laser therapy can significantly enhance and accelerate this process.
- Be Precise with Your Diagnosis: Vague diagnoses lead to poor outcomes. Differentiating between a meniscal repair and a meniscectomy, for example, is critical to understanding the patient’s current pathology and formulating the correct treatment plan. The treatment must match the diagnosis.
Disclaimer: This educational post is intended for informational purposes only and is based on the clinical experience and research interpretation of Dr. Alexander Jimenez, DC, FNP-APRN. The information provided is not intended to be a substitute for professional medical advice, diagnosis, or treatment. It should not be used as medical advice for your specific situation.
Disclaimer for Individuals: All individuals should seek the advice of their own physician or another qualified health provider with any questions they may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this web page. You must obtain recommendations for your personal situation from your own medical providers.
References:
- Farr, J., Prat, D., & Lattermann, C. (2020). State of the Art in Biologics for Cartilage Repair. Springer. (Conceptual basis for biologics in cartilage).
- Laudy, A. B., Bakker, E. W., Rekers, M., & Moen, M. H. (2015). Efficacy of platelet-rich plasma injections in osteoarthritis of the knee: a systematic review and meta-analysis. British Journal of Sports Medicine, 49(10), 657-665.
- Mautner, K., Blazuk, J., & Shiple, B. (2018). The Role of Alpha-2-Macroglobulin in the Treatment of Osteoarthritis. Current Physical Medicine and Rehabilitation Reports, 6(1), 1-6.
- O’Connell, B., Wragg, N. M., & Wilson, S. L. (2019). The use of platelet-rich plasma in musculoskeletal pathology. Journal of Bone and Joint Surgery. British volume, 101(1), 8-15.
- Hamblin, M. R. (2017). Mechanisms and applications of the anti-inflammatory effects of photobiomodulation. AIMS biophysics, 4(3), 337.
- Cassano, P., Petrie, S. R., Mischoulon, D., et al. (2018). The Neuroscience of Low-level Light Therapy. Annals of the New York Academy of Sciences, 1431(1), 108-120.
- Englund, M., Guermazi, A., & Lohmander, L. S. (2009). The role of meniscectomy in the development of knee osteoarthritis. Rheumatic Disease Clinics, 35(3), 577-587.
Keywords: Platelet-Rich Plasma, PRP, Protein Concentrate, Platelet-Poor Plasma, PPP, Orthobiologics, Regenerative Medicine, Dr. Alexander Jimenez, Osteoarthritis, Platelet Dose, Platelet Recovery, Alpha-2-Macroglobulin, A2M, Interleukin-1 Receptor Antagonist, IL-1ra, Ultrasound-Guided Injection, Acromioclavicular Joint, AC Joint, Photobiomodulation, Laser Therapy, Meniscectomy, Knee Pain, Shoulder Pain, Rehabilitation.
General Disclaimer
Professional Scope of Practice *
The information herein on "Photobiomodulation Techniques You Need for Precision PRP" is not intended to replace a one-on-one relationship with a qualified health care professional or licensed physician and is not medical advice. We encourage you to make healthcare decisions based on your research and partnership with a qualified healthcare professional.
Blog Information & Scope Discussions
Welcome to El Paso's Premier Wellness and Injury Care Clinic & Wellness Blog, where Dr. Alex Jimenez, DC, FNP-C, a Multi-State board-certified Family Practice Nurse Practitioner (FNP-BC) and Chiropractor (DC), presents insights on how our multidisciplinary team is dedicated to holistic healing and personalized care. Our practice aligns with evidence-based treatment protocols inspired by integrative medicine principles, similar to those found on this site and our family practice-based chiromed.com site, focusing on restoring health naturally for patients of all ages.
Our areas of multidisciplinary practice include Wellness & Nutrition, Chronic Pain, Personal Injury, Auto Accident Care, Work Injuries, Back Injury, Low Back Pain, Neck Pain, Migraine Headaches, Sports Injuries, Severe Sciatica, Scoliosis, Complex Herniated Discs, Fibromyalgia, Chronic Pain, Complex Injuries, Stress Management, Functional Medicine Treatments, and in-scope care protocols.
Our information scope is multidisciplinary, focusing on musculoskeletal and physical medicine, wellness, contributing etiological viscerosomatic disturbances within clinical presentations, associated somato-visceral reflex clinical dynamics, subluxation complexes, sensitive health issues, and functional medicine articles, topics, and discussions.
We provide and present clinical collaboration with specialists from various disciplines. Each specialist is governed by their professional scope of practice and their jurisdiction of licensure. We use functional health & wellness protocols to treat and support care for musculoskeletal injuries or disorders.
Our videos, posts, topics, and insights address clinical matters and issues that are directly or indirectly related to our clinical scope of practice.
Our office has made a reasonable effort to provide supportive citations and has identified relevant research studies that support our posts. We provide copies of supporting research studies upon request to regulatory boards and the public.
We understand that we cover matters that require an additional explanation of how they may assist in a particular care plan or treatment protocol; therefore, to discuss the subject matter above further, please feel free to ask Dr. Alex Jimenez, DC, APRN, FNP-BC, or contact us at 915-850-0900.
We are here to help you and your family.
Blessings
Dr. Alex Jimenez DC, MSACP, APRN, FNP-BC*, CCST, IFMCP, CFMP, ATN
email: coach@elpasofunctionalmedicine.com
Multidisciplinary Licensing & Board Certifications:
Licensed as a Doctor of Chiropractic (DC) in Texas & New Mexico*
Texas DC License #: TX5807, Verified: TX5807
New Mexico DC License #: NM-DC2182, Verified: NM-DC2182
Multi-State Advanced Practice Registered Nurse (APRN*) in Texas & Multistate
Multistate Compact RN License by Endorsement (42 States)
Texas APRN License #: 1191402, Verified: 1191402 *
Florida APRN License #: 11043890, Verified: APRN11043890 *
* Prescriptive Authority Authorized
ANCC FNP-BC: Board Certified Nurse Practitioner*
Compact Status: Multi-State License: Authorized to Practice in 40 States*
Graduate with Honors: ICHS: MSN-FNP (Family Nurse Practitioner Program)
Degree Granted. Master's in Family Practice MSN Diploma (Cum Laude)
Dr. Alex Jimenez, DC, APRN, FNP-BC*, CFMP, IFMCP, ATN, CCST
My Digital Business Card
RN: Registered Nurse
APRNP: Advanced Practice Registered Nurse
FNP: Family Practice Specialization
DC: Doctor of Chiropractic
CFMP: Certified Functional Medicine Provider
MSN-FNP: Master of Science in Family Practice Medicine
MSACP: Master of Science in Advanced Clinical Practice
IFMCP: Institute of Functional Medicine
CCST: Certified Chiropractic Spinal Trauma
ATN: Advanced Translational Neutrogenomics